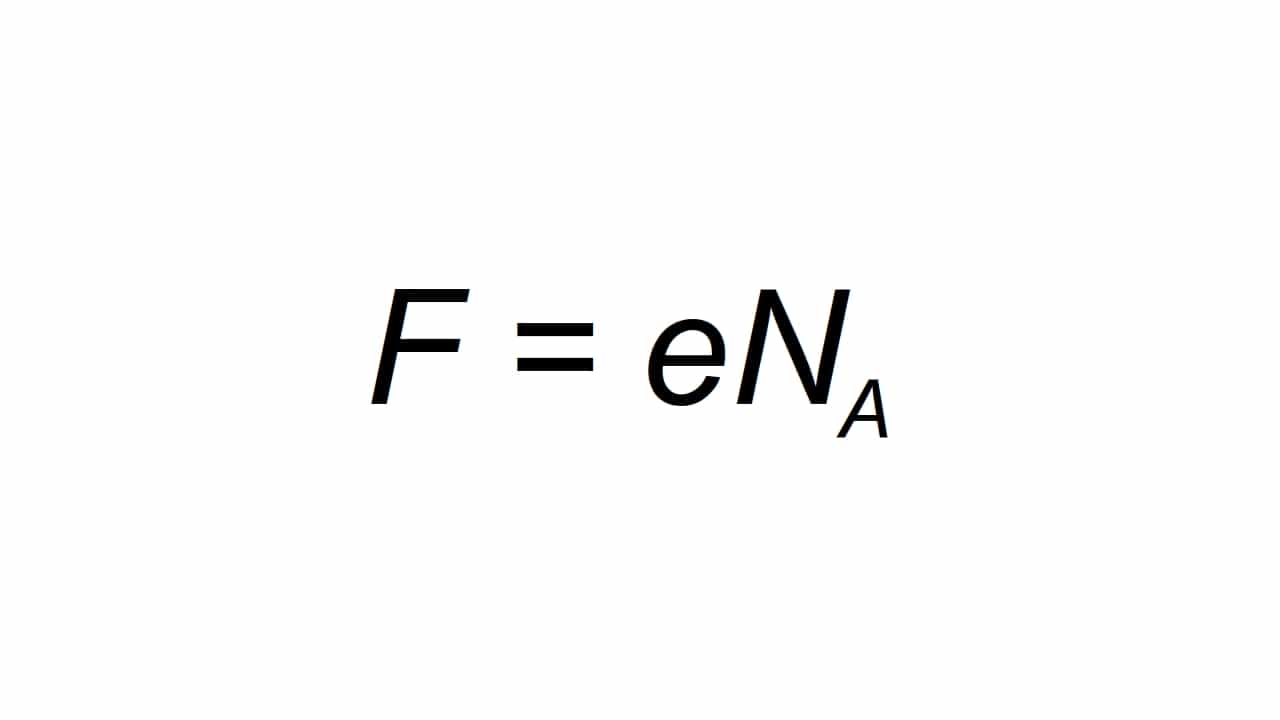Sedzimwe nguva isu takataura pane mimwe mibvunzo yakakosha mumunda wemagetsi nemagetsi, senge Mutemo waOmm, masaisai Kirchoff mitemo, uye kunyange iyo mhando dzemaseketi emagetsi akakosha, zvinganakidzewo kuziva kuti chii Kugara kwaFaraday, sezvo inogona kukubatsira iwe kuziva zvishoma zvishoma nezve iyo mitoro.
Muchikamu chino iwe unonzwisisa zvishoma zvirinani chii chinogara chiri mufaro, chingaiswa kupi, uye chinoverengerwa sei ...
Chii chinonzi Faraday chinogara chiripo?
La Kugara kwaFaraday inogara ichishandiswa zvakanyanya muminda yefizikisi uye kemesitiri. Inotsanangurwa sehuwandu hwemagetsi emagetsi pa mole yemagetsi. Zita rayo rinobva kuna musayendisiti weBritish Michael Faraday. Izvi zvinogara zvichishandiswa mumagetsi emagetsi kuongorora huwandu hwezvinhu zvinoumba mu electrode.
Inogona kumiririrwa netsamba F, uye inotsanangurwa seye molar elemental kuchaja, kuve uchikwanisa kuverenga akadai se:
Kuva F kukosha kunoguma yekugara kwaFarday, e iyo yekutanga element magetsi, uye Na ndeye Avogadro anogara aripo:
- e = 1.602176634 × 10-19 C
- Na = 6.02214076 × 1023 at the mole-1
Zvinoenderana neSI iyi Faraday inogara iri chaiyo, semamwe maumbirwo, uye kukosha kwayo chaiko kuri: 96485,3321233100184 C / mol. Sezvauri kuona, inoratidzwa mune yeyuniti C / mol, ndokuti, coulombs pa mole. Uye kuti unzwisise kuti izvi zvikamu ndezvipi, kana usati waziva, unogona kuenderera kuverenga zvikamu zviviri zvinotevera ...
Chii chinonzi mole?
Un at the mole chinhu chinoyera huwandu hwezvinhu. Mukati meSI yemayuniti, ndeimwe yeanokosha e7. Mune chero chinhu, chingave chinhu kana chemakemikari chinoumbwa, pane akateedzana ezvikamu zvekutanga izvo zvinoumba. Rimwe mole rinenge rakaenzana ne6,022 140 76 × 1023 zvinhu zvekutanga, inova iyo yakasarudzika manhamba kukosha kweiyo Avogadro inogara iripo.
Izvi zvinhu zvekutanga zvinogona kuve atomu, morekuru, ion, elektroni, mafotoni, kana chero imwe mhando yechinhu chepasi. Semuenzaniso, neizvi unogona Verenga huwandu hweatomu chii chiri mugiramu yechinhu chakapihwa.
In the chemistry, iyo mole yakakosha, nekuti inobvumidza kuwanda kuti kuitwe kwekuumbwa, kusangana kwemakemikari, nezvimwe. Semuenzaniso, yemvura (H2O), iwe une maitiro 2 H2 +O2 → 2H2O, ndiko kuti, ma moles maviri ehydrogen (H2) uye rimwe mole reoksijeni (O2) ita kuti uite mafomu maviri emvura. Uyezve, dzinogona zvakare kushandiswa kuratidza kusangana (ona kuwanda).
Chii chinonzi mari yemagetsi?
Kune rimwe divi, kubva ku mutero wemagetsi Isu tato taura pane dzimwe nguva, inzvimbo yepanyama yezvimwe zvikamu zveatomic zvinoratidza masimba anoyevedza uye anosemesa pakati pavo nekuda kwemagetsi emagetsi. Kubatana kwemagetsi, pakati pechaji nemunda wemagetsi, ndeimwe yezvinhu zvina zvakakosha zvefizikiki, pamwe neiyo simba renyukireya, simba renyukireya risina simba, uye simba rekukweva.
Kuyera mari yemagetsi iyi, iyo Coulomb (C) kana Coulomb, uye inotsanangurwa sehuwandu hwebhadharo hunotakurwa mune imwe sekondi nemagetsi emagetsi ekuwedzera one ampere.
Zvishandiso zveiyo Faraday zvinogara

Kana iwe uchinetseka kuti chii inoshanda kushanda Unogona kuve neichi chinogara chiri Faraday, chokwadi ndechekuti iwe une akati wandei, mimwe mienzaniso ndeiyi:
- Electroplating / anodizing: yezvakaitwa mu metallurgia indasitiri uko imwe simbi yakafukidzwa neimwe ne electrolysis. Semuenzaniso, kana simbi yakakwenenzverwa nechero zingi kuti ipe kukurira kurwisa. Mune maitirwo aya, simbi inoputirwa inoshandiswa seanode uye iyo electrolyte isunyu yakanyungudika yezvinhu zveanode.
- Simbi yekuchenesa: iyo inogona zvakare kushandiswa kune mafomula anoshandiswa pakunatsiridza simbi senge mhangura, zinc, tini, nezvimwe. Zvakare nemaelectrolysis maitiro.
- Chemical kugadzira: kuburitsa makemikari makemikari ichi chinogara chichiwanzo shandiswa.
- Chemical kuongorora: ne electrolysis iyo makemikari akaumbwa anogona zvakare kusarudzwa.
- Kugadzirwa kwegesi: magasi akadai seoksijeni kana hydrogen anowanikwa kubva mumvura neelectrolysis anoshandisawo izvi nguva dzese pakuverenga.
- Mishonga uye aestheticsElectrolysis inogona zvakare kushandiswa kumutsa dzimwe tsinga kana kurapa mamwe matambudziko, mukuwedzera pakubvisa bvudzi risingadiwe. Pasina zvinogara zviripo, kuwanda kwematurusi erudzi urwu kwaigona kunge kusiri kugadzirwa.
- Printing services: Kune maprinta, maitiro e electrolysis anoshandiswawo kune zvimwe zvinhu.
- Electrolytic capacitors: inozivikanwa zvemagetsi chinhu chine mutete firimu yealuminium oxide uye aluminium anode pakati pemagetsi. Iyo electrolyte musanganiswa we boric acid, glycerin, uye ammonium hydroxide. Uye aya ndiwo mabudiro anoita iwo makuru masimba ...
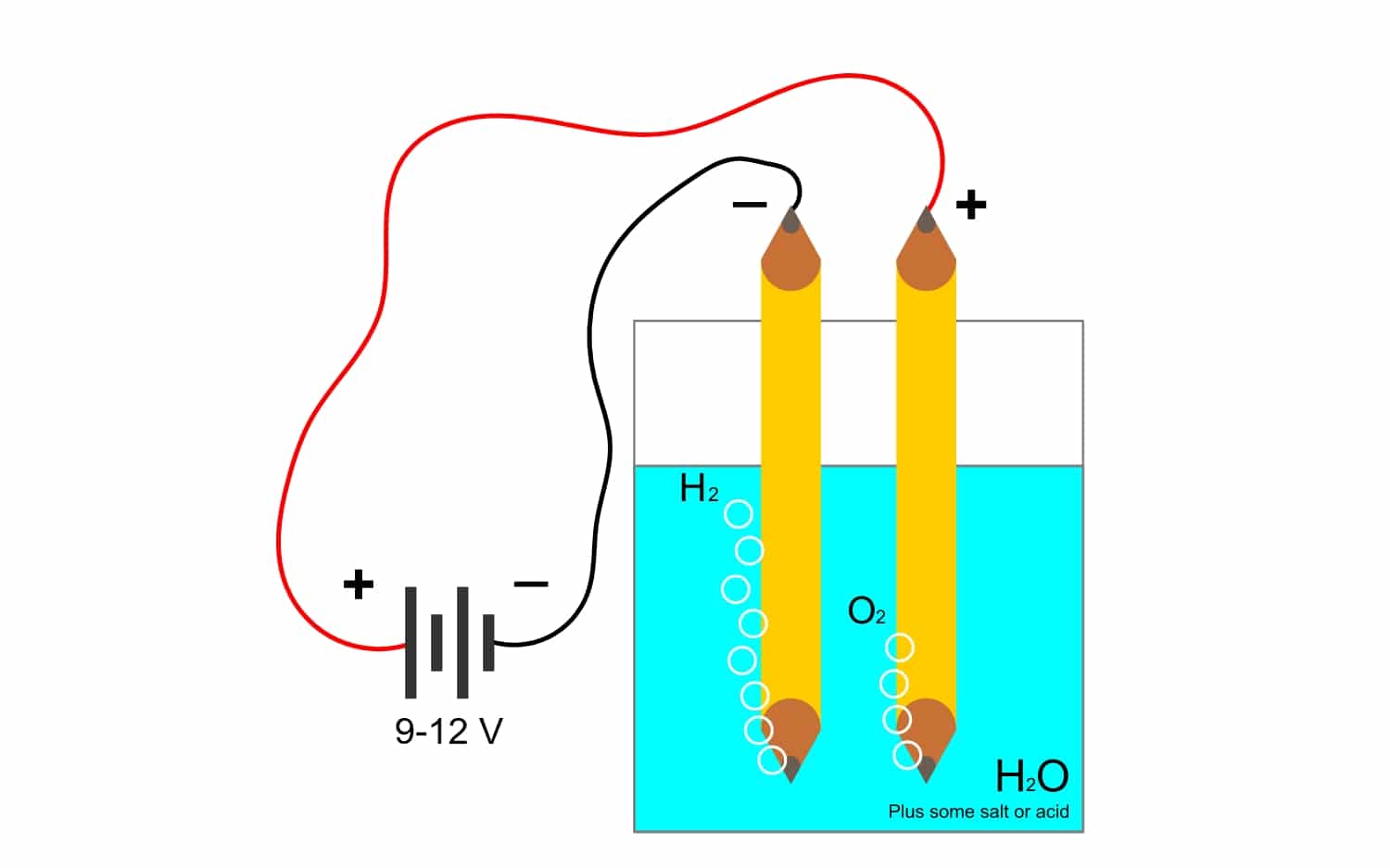
Chii chinonzi electrolysis?
Uye sezvo kugara kweFaraday kwakabatana zvakanyanya neiyo electrolysisNgatione kuti iri rimwe izwi nderei rinoshandiswa zvakanyanya muindasitiri. Kutenda nemaitiro aya, zvinhu zvemubatanidzwa zvinogona kupatsanurwa nemagetsi. Izvi zvinoitwa nekuburitswa kwemaerekitironi neanode anions (oxidation) uye kubatwa kwemaerekitironi nekatodhi cations (kudzikisira).
Yakawanikwa netsaona naWilliam Nicholson, muna 1800, apo aidzidza mashandiro emakemikari mabhatiri. Muna 1834, Michael Faraday yakagadzira uye yakaburitsa iyo mitemo ye electrolysis.
Semuenzaniso, electrolysis ye mvura H2O, inobvumira kugadzira oxygen uye hydrogen. Kana yakananga yazvino ikaiswa kuburikidza nemagetsi, ayo anoparadzanisa okisijeni kubva kuhydrogen, uye nekugona kupatsanura magasi ese ari maviri (haakwanise kusangana, nekuti anounza chinhu chinotyisa chinoputika).